ICH E9(R1) 临床试验中的估计目标与敏感性分析(E9指导原则增补文件)
English Version
ICH E9(R1) Addendum: Statistical Principles for Clinical TrialsA.1. 目的和范围
为了给制药公司、监管机构、患者、医生和其他利益相关方的决策提供正确的信息,应明确描述特定医疗条件下治疗(药物)的获益和风险。如果不能对此进行明确描述,报告的“治疗效应”可能会被误解。本增补提出了一个结构化的框架,以加强参与制定临床试验目的、设计、实施、分析和解释的多学科间的交流,并加强申办方和监管机构之间关于临床试验中治疗效应的沟通。
构建相应临床问题的“估计目标”(见词汇表;A.3.)有助于精确描述治疗效应,这就需要深思熟虑地定义“伴发事件” (见词汇表;A.3.1.),如终止分配的治疗,使用额外或其他治疗,或终末事件(如死亡)等。估计目标的描述应该反映出与这些伴发事件相关的临床问题,并且本增补介绍了反映不同临床问题的策略。在描述临床问题时,策略的选择可能会影响到如何反映试验的更加常规的属性,例如治疗、人群或相关的变量(终点)。
临床试验数据的统计分析应当与估计目标对应。本增补阐明了“敏感性分析”(见词汇表)在探索主要统计分析结论稳健性中的作用。
本增补中,对原始ICH E9的引用采用x.y格式,对本增补的引用采用A.x.y.格式。
本增补就以下若干方面澄清和扩展了ICH E9。第一,ICH E9介绍了随机对照试验中对应于疗法策略的意向治疗(ITT)原则,据此对受试者进行随访、评估和分析,而不考虑其是否依从计划的治疗过程,这表明保持随机化为统计学检验提供了一个坚实的基础。ITT 原则具有以下三个含义。首先,试验分析应包括与研究问题相关的所有受试者。其次,受试者应按随机化时的分配纳入分析。最后,根据ITT原则(见ICH E9词汇表)的定义,无论是否依从预定的治疗过程,都应对受试者进行随访和评估,并在分析中使用这些评估。毫无疑问,随机化是对照临床试验的基石,分析时应最大限度地利用随机化的这一优势。然而,根据ITT 原则估计治疗效应能否总是代表与监管和临床决策最相关的治疗效应,这个问题仍然悬而未决。本增补中概述的框架为描述不同的治疗效应提供了基础,并提出了试验设计和分析需考虑的要点,以便估计治疗效应,为决策提供可靠依据。
第二,本增补重新审视了通常归为数据处理和“缺失数据”(见词汇表)的一些问题,并提出了两个重要的区别。首先,增补对终止随机分配的治疗和退出研究加以区分。前者代表一个伴发事件,需通过在试验目的中对估计目标的精确说明加以解决;后者导致缺失数据,需在统计分析中加以解决。例如,考虑在肿瘤学试验中转组治疗的受试者,以及由于试验完成而无法观测到结局事件的受试者。前者代表伴发事件,关于该事件的临床问题应明确。后者属于管理性删失,需要在统计分析中作为缺失数据问题加以解决。估计目标的清晰性为计划需要收集哪些数据提供了依据,以及哪些数据如果未被收集到即为缺失数据问题,需要在统计分析中加以解决。然后,可以选择解决缺失数据问题的方法,以与估计目标一致。其次,增补强调了不同伴发事件的不同影响。诸如终止治疗、转组治疗或使用额外药物等事件可能导致变量的后续观测值即使可以收集到数据也与估计目标不相关或难以解释。而对于死亡的受试者,死亡后的观测值是不存在的。
第三,在框架中考虑了与分析集概念相关的问题。第 5.2.节强烈建议优效性试验的分析基于全分析集,即尽可能包括所有随机化受试者的分析集。然而,试验往往包括对同一受试者的重复观测。某些受试者按计划收集的观测值可能被认为是无关的或难以解释的,剔除这些观测值,与从全分析集中完全剔除受试者可能具有类似的后果,即没有完全保留最初的随机化。这样做的一个后果是,随机化赋予关于治疗效应的检验假设的理论优势获益以及平衡基线混杂因素的实际获益可能被削弱。另外,有意义的结局变量取值可能不存在,例如当受试者已死亡。第 5.2.节没有直接阐明这些问题。这些问题要在考虑伴发事件的前提下,通过仔细定义关注的治疗效应来进行明确,既要确定要包括在治疗效应估计中的受试者人群,又要确定每个受试者包括在分析中的观测值。本增补也重新审视了使用符合方案集来分析的意义和作用,尤其是,是否需要用比分析符合方案集更能减少偏倚、更有可解读性的方式,来研究方案违背和偏离的影响。
最后,在敏感性分析部分进一步讨论了稳健性的概念(见1.2.)。特别区分了所选分析方法的假设的敏感性,以及分析方法选择上的敏感性。通过精确说明已达成共识的估计目标,以及与估计目标一致的分析方法且其预先设定的细节描述达到能使第三方精确地重现分析结果的程度,这样,监管机构对于一个特定分析可聚焦于假设偏离和数据局限的敏感性。
无论是基于有效性或安全性的治疗效应估计,还是对治疗效应相关假设的检验,本增补中概述的原则均适用。虽然主要关注的是随机临床试验,但这些原则也同样适用于单臂试验和观察性研究。该框架适用于任何数据类型,包括纵向数据、首次事件发作时间数据和复发事件数据。对于确证性临床试验和用于产生确证性结论的跨试验整合数据,监管部门对所述原则的应用将更为关注。
A.2. 将计划、设计、实施、分析和解释协调一致的框架
试验计划应按顺序进行(图 1)。应通过定义合适的估计目标,将明确的试验目的转化为关键的临床所关注的问题。估计目标根据特定的试验目的定义估计的目标(即“要估计什么”,见A.3.),然后可以选择合适的估计方法(即分析方法,称为主“估计方法”,见词汇表)(见 A.5.1.)。主估计方法将以特定假设为基础,为了探索根据主估计方法所作推断对偏离其基本假设的稳健性,应针对同一估计目标采用一种或多种形式进行敏感性分析(见A.5.2.)。
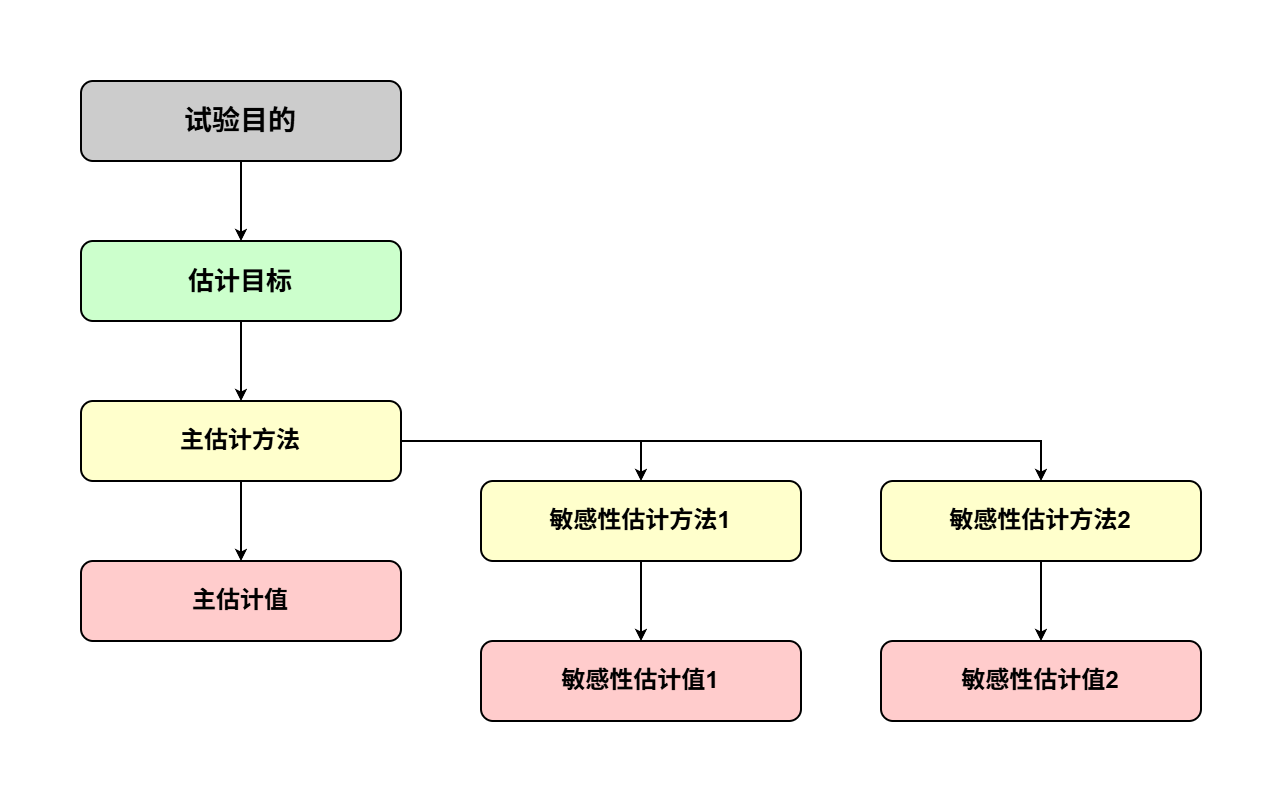
图1:协调估计的目标、估计的方法和敏感性分析,使其与给定试验目的对应
该框架有助于制定适当的试验计划,以明确区分估计的目标(试验目的,估计目标)、估计的方法(估计方法)、数值结果(“估计值”,见词汇表)和敏感性分析。这将有助于申办方的试验计划制定和监管机构的审评工作,并在双方讨论临床试验设计的适宜性和临床试验结果的解释时增强交流。
指定适当的估计目标(见 A.3.)通常是试验设计、实施(见A.4.)和分析(见A.5.)方面的主要决定因素。
A.3. 估计目标
药物开发和批准的核心问题是明确治疗效应是否存在,并估计其大小:如何比较相同受试者接受不同治疗的结局(即,如果受试者未接受治疗或接受不同治疗)。估计目标是对治疗效应的精确描述,反映了既定临床试验目的提出的临床问题。它在群体层面上总结了同一批患者在不同治疗条件下比较的结果。估计的目标将在临床试验之前定义。一旦定义了估计的目标,即可设计试验以可靠地估计治疗效应。
估计目标的描述涉及特定属性的精确说明,这些属性不仅应基于临床考虑而制定,还应基于所关注的临床问题中如何反映伴发事件。第 A.3.1.节介绍了伴发事件。第 A.3.2.节介绍了各种策略,来描述与伴发事件有关的问题。第 A.3.3.节描述了估计目标的属性,第 A.3.4.节则提出了估计目标构建的考虑要点。理解不同策略之间的差异,并精确阐明哪些策略用于构建估计目标,这一点至关重要。
A.3.1. 临床问题中反映的伴发事件
伴发事件是指治疗开始后发生的事件,可影响与临床问题相关的观测结果的解读或存在。在描述临床问题时,有必要阐明伴发事件,以便准确定义需要估计的治疗效应。
在描述治疗效应时需要考虑伴发事件,因为变量的观测结果可能受伴发事件的影响,而伴发事件的发生可能取决于治疗。例如,两名患者可能最初暴露于相同的治疗并提供相同的结局观测值,但如果其中一名患者接受了其他药物治疗,则两名患者之间,观测值所反映的治疗的信息会有所不同。此外,患者接受的治疗会影响到他们是否需要服用其他用药,以及是否可以继续接受治疗。与缺失数据不同,伴发事件不应被认为是临床试验中需要避免的缺陷。在临床试验中发生的终止既定治疗、使用其他药物和其他此类事件在临床实践中也可能发生,因此在定义临床问题时需要明确考虑这些事件发生的可能。
可影响观测结果解释的伴发事件包括终止所分配的治疗,和使用额外或其他治疗。使用额外或其他疗法可以有多种形式,包括改变基础治疗或合并治疗、转组治疗。影响观测结果存在的伴发事件包括终末事件,例如死亡和腿截肢(当评估糖尿病性足溃疡的症状时),而且这些事件不是变量本身的一部分。当某些临床事件的发生或不发生定义了一个主层时(见 A.3.2.),这些事件也可以是伴发事件。例如, 肿瘤领域中在评估缓解持续时间疗效时定义客观缓解的肿瘤缩小;对于初始未感染的接种疫苗受试者在评估感染严重程度疗效时的感染发生。
伴发事件可能仅由事件本身确定,如终止治疗,或可能有更详细的定义。详细的定义例如,可明确说明事件发生的原因,如因毒性作用终止治疗,或因缺乏疗效而终止治疗;事件可能需要达到一定量级或程度,如使用超过规定时间或剂量的其他药物;或明确说明事件发生的时机,可能与其对变量评估的接近程度有关。一些事件会无限期地影响结局观测值的解释,例如终止治疗,而另一些事件只会暂时影响,例如短期使用其他治疗。事实上,额外或其他治疗可以是多样的;可以是替代或补充受试者获益不足时的治疗,或作为对既定治疗不耐受的另一种选择,或作为控制疾病暂时急性发作的短期急性治疗。在临床试验中,额外或其他治疗通常是指诸如基础治疗、补救药物和禁用药物,要区分它们的不同作用以对其分别考虑。如果要使用不同的策略,则需要额外的详细信息,确定不同的伴发事件。例如,如果伴发事件不仅取决于未继续治疗,还取决于与未继续治疗相关的原因、程度或时机,则应在临床试验中准确定义和记录该附加信息。理论上,描述伴发事件可能体现治疗和随访非常具体的细节,例如长期治疗的单次漏服或日间服药的错误时间。如果预期这些具体标准不会影响对变量的解释,则不需要将它们作为伴发事件处理。
如上所述,在构建估计目标时需要考虑伴发事件。因为估计目标要在试验设计之前进行定义,所以无论是退出研究还是其他缺失数据的原因(例如生存结局的试验中的管理性删失)本身都不是伴发事件。退出试验的受试者在退出前可能已经发生了伴发事件。
A.3.2. Strategies for Addressing Intercurrent Events when Defining the Clinical Question of Interest 在定义临床问题时解决伴发事件的策略
Descriptions of various strategies are listed below, each reflecting a different clinical question of interest in respect of a particular intercurrent event. Whether or not the naming convention is used, it is required that the choices of strategy are unambiguously clear once the estimand is constructed. It is not necessary to use the same strategy to address all intercurrent events. Indeed, different strategies will often be used to reflect the clinical question of interest in respect of different intercurrent events. Section下面列出了多种策略,每种策略又体现了对于特定伴发事件的不同临床问题。无论是否使用如下命名规则,构建估计目标时策略的选择都必须清晰明确。无需使用相同的策略来处理所有的伴发事件。事实上,通常会使用不同的策略来明确体现不同伴发事件的临床问题。第 A.3.4. gives some considerations on selecting strategies to construct an estimand.节给出了一些在构建估计目标时策略选择上的考虑。
Treatment policy strategy 疗法策略
The疗法策略下伴发事件的发生与定义治疗效应无关,即无论是否发生伴发事件,均会使用相关变量的值。例如,将使用其他药物治疗作为伴发事件时,规定无论患者是否服用其他药物,都使用相关变量的值。
对于患者是否继续治疗以及患者的其他治疗(基础或合并治疗)是否有变化等伴发事件,在疗法策略中被视为治疗的一部分。基于这种情况的比较就体现了ICH ofE9所阐述的ITT原则,比较结果亦是疗法策略下的治疗效应。
一般情况下,对于终末事件类型的伴发事件,不能采用疗法策略,原因是该类伴发事件后变量的值不再存在。例如,在死亡之后变量是无法观测的,因此不能基于此策略构建估计目标。
假想策略
假想策略设想一种没有发生伴发事件的情景:此时,体现临床问题的变量值是在所假设的情景下采用的变量值。
存在各种各样的假设情景,但其中有些情景更具临床或监管意义。例如,在与可实施试验条件不同的条件下的治疗效应可能具有临床或监管重要性。具体而言,当出于伦理原因必须提供额外药物治疗时,可能要考虑未提供额外药物情形下的治疗效应。一个非常不同的假设情景可能是假定伴发事件不会发生,或者会发生不同的伴发事件。例如,对于因发生不良事件而终止治疗的受试者,可考虑同一受试者没有发生不良事件或即使发生不良事件仍然继续治疗的情景。这种假设情景的临床和监管意义有限,并且通常需要清楚地理解伴发事件或其后果在临床实践与临床试验中为什么不同以及如何不同。
如果提出了一个假想策略,应该明确具体的假设情景是什么。举例来说,诸如“如果患者未服用额外药物”之类的措辞可
irrelevant能会导致混淆, in因为不清楚患者是因为没有额外药物可用而未服用,还是该患者不需要服用额外药物而未服用。
复合变量策略
复合变量策略与关注的变量有关(见 treatmentA.3.3.)。伴发事件本身可提供关于患者结局的信息,因此将其纳入变量的定义之中。例如,由于毒性而终止治疗的患者可能被认为治疗失败。如果变量已被定义为成功或失败,因毒性终止治疗将被认为是另一种形式的失败。复合变量策略不仅限于二分类变量,也可以是连续型变量。例如,在观测生理功能的试验中,死亡的受试者可以用某一数值代表生理功能缺失。当变量原始观测值可能不存在或没有意义,但是伴发事件本身能够体现患者结局(如患者死亡)时,可将复合变量策略视为遵循意向治疗原则的策略。
终末事件,如死亡,可能是需要采用复合策略的最突出例子。如果某种治疗可以挽救生命,可能会关注其对存活患者的各种指标的作用, of但是,如果汇总指标仅关注存活患者的一些数值指标的平均值是不够的,要同时关注数值指标和是否生存。例如,肿瘤试验中的无进展生存期衡量了肿瘤生长和生存组合在一起的治疗效应。
在治策略
在治策略关注在伴发事件发生之前的治疗效应。该策略的具体术语将取决于相关伴发事件;例如,当将死亡视为伴发事件时,可以称为“在世策略”。
如果一个变量被重复测量,则伴发事件发生前的所有观测值都可能被认为与临床问题相关,而不是所有受试者在相同固定时间点的值。这也适用于二分类结局在伴发事件之前发生的情况。例如,处于终末期的受试者可能会因为死亡而终止对症治疗,但可以根据死亡前症状的缓解情况评估治疗效果。还有一种情形,受试者可能终止治疗,此时评估其暴露于治疗期间药物不良反应的风险是值得关注的。
因此,在治策略与复合变量策略类似,会影响变量的定义。在这种情况下,在治策略通过将相应的观测时间限制在伴发事件之前来影响。如果各治疗组间的伴发事件的发生率不同,则尤其需要谨慎(见A.3.3.)。
主层策略
主层策略与人群有关(见 of interest is used regardless of whether or not the intercurrent event occurs. For example, when specifying how to address use of additional medication as an intercurrent event, the values of the variable of interest are used whether or not the patient takes additional medication.A.3.3.)。可认为目标人群是会发生伴发事件的“主层”(见词汇表)。或者,目标人群是不会发生伴发事件的主层。临床问题仅在该主层中与治疗效应相关。例如,在接种疫苗后仍然感染的患者主层中,可能需要了解针对感染严重程度的治疗效应。或者,毒性可能会使一些患者无法继续接受试验药物,但需要了解能够耐受试验药物的患者的治疗效应。
If applied in relation to whether or not a patient continues treatment, and whether or not a patient experiences changes in other treatments (e.g. background or concomitant treatments), the intercurrent event is considered to be part of the treatments being compared. In that case, this reflects the comparison described in the ICH E9 Glossary (under ITT Principle) as the effect of a treatment policy.
In general, the treatment policy strategy cannot be implemented for intercurrent events that are terminal events, since values for the variable after the intercurrent event do not exist. For example, an estimand based on this strategy cannot be constructed with respect to a variable that cannot be measured due to death.
Hypothetical strategies
A scenario is envisaged in which the intercurrent event would not occur: the value of the variable to reflect the clinical question of interest is the value which the variable would have taken in the hypothetical scenario defined.
A wide variety of hypothetical scenarios can be envisaged, but some scenarios are likely to be of more clinical or regulatory interest than others. For example, it may be of clinical or regulatory importance to consider the effect of a treatment under different conditions from those of the trial that can be carried out. Specifically, when additional medication must be made available for ethical reasons, a treatment effect of interest might concern the outcomes if the additional medication was not available. A very different hypothetical scenario might postulate that intercurrent events would not occur, or that different intercurrent events would occur. For example, for a subject that will suffer an adverse event and discontinue treatment, it might be considered whether the same subject would not have the adverse event or could continue treatment in spite of the adverse event. The clinical and regulatory interest of such hypotheticals is limited and would usually depend on a clear understanding of why and how the intercurrent event or its consequences would be expected to be different in clinical practice than in the clinical trial.
If a hypothetical strategy is proposed, it should be made clear what hypothetical scenario is envisaged. For example, wording such as 区分“主层”和子集很重要。“if the patient does not take additional medication” might lead to confusion as to whether the patient hypothetically does not take additional medication because it is not available or because the particular patient is supposed not to require it.
Composite variable strategies
This relates to the variable of interest (see A.3.3.). An intercurrent event is considered in itself to be informative about the patient’s outcome and is therefore incorporated into the definition of the variable. For example, a patient who discontinues treatment because of toxicity may be considered not to have been successfully treated. If the outcome variable was already success or failure, discontinuation of treatment for toxicity would simply be considered another mode of failure. Composite variable strategies do not need to be limited to dichotomous outcomes, however. For example, in a trial measuring physical functioning, a variable might be constructed using outcomes on a continuous scale, with subjects who die being attributed a value reflecting the lack of ability to function. Composite variable strategies can be viewed as implementing the intention-to-treat principle in some cases where the original measurement of the variable might not exist or might not be meaningful, but where the intercurrent event itself meaningfully describes the patient’s outcome, such as when the patient dies.
Terminal events, such as death, are perhaps the most salient examples of the need for the composite strategy. If a treatment saves lives, its effect on various measures in surviving patients may be of interest, but it would be inappropriate to say that the summary measure of interest was only the average value of some numerical measure in survivors. The outcome of interest is survival along with the numerical measures. For example, progression-free survival in oncology trials measures the treatment effect on a combination of the growth of the tumour and survival.
While on treatment strategies
For this strategy, response to treatment prior to the occurrence of the intercurrent event is of interest. Terminology for this strategy will depend on the intercurrent event of interest; e.g. “while alive”, when considering death as an intercurrent event.
If a variable is measured repeatedly, its values up to the time of the intercurrent event may be considered relevant for the clinical question, rather than the value at the same fixed timepoint for all subjects. The same applies to the occurrence of a binary outcome of interest up to the time of the intercurrent event. For example, subjects with a terminal illness may discontinue a purely symptomatic treatment because they die, yet the success of the treatment can be measured based on the effect on symptoms before death. Alternatively, subjects might discontinue treatment and, in some circumstances, it will be of interest to assess the risk of an adverse drug reaction while the patient is exposed to treatment.
Like the composite variable strategy, the while on treatment strategy can hence be thought of as impacting the definition of the variable, in this case by restricting the observation time of interest to the time before the intercurrent event. Particular care is required if the occurrence of the intercurrent event differs between the treatments being compared (see A.3.3.).
Principal stratum strategies
This relates to the population of interest (see A.3.3.). The target population might be taken to be the “principal stratum” (see Glossary) in which an intercurrent event would occur. Alternatively, the target population might be taken to be the principal stratum in which an intercurrent event would not occur. The clinical question of interest relates to the treatment effect only within the principal stratum. For example, it might be desired to know a treatment effect on severity of infections in the principal stratum of patients becoming infected after vaccination. Alternatively, a toxicity might prevent some patients from continuing the test treatment, but it would be desired to know the treatment effect among patients who are able to tolerate the test treatment.
It is important to distinguish “principal stratification” (see Glossary), which is based on potential intercurrent events (for example, subjects who would discontinue therapy if assigned to the test product), from subsetting based on actual intercurrent events (subjects who discontinue therapy on their assigned treatment). The subset of subjects who experience an intercurrent event on the test treatment will often be a different subset from those who experience the same intercurrent event on control. Treatment effects defined by comparing outcomes in these subsets confound the effects of the different treatments with the differences in outcomes possibly due to the differing characteristics of the subjects.主层”(见词汇表)是基于潜在的伴发事件(例如,若分配到试验组将终止治疗的受试者),而“子集”是基于实际发生的伴发事件(终止既定治疗的受试者)。在试验组发生伴发事件的受试者子集通常与对照组发生相同伴发事件的受试者子集不同。比较这些子集的结局而定义的治疗效应,会混杂不同治疗间的真实效应和可能由于受试者不同特征导致的结局差异。
A.3.3. Estimand Attributes估计目标的属性
The attributes below are used to construct the estimand, defining the treatment effect of interest. 下述属性用于构建估计目标,定义相关的治疗效应。
The治疗(处理):相关的治疗条件,以及适用时进行比较的其他治疗条件(在本文件其余部分中称为“治疗”)。这些可能是单独的干预措施,也可能是同时进行的干预措施的组合(例如作为加载治疗),或者是一个复杂干预序列组成的整体方案。(请见A.3.2.下的疗法策略和假想策略)。
人群:临床问题所针对的患者人群。可以是整个试验人群,也可以是按某种基线特征定义的亚组,或由特定伴发事件的发生(或不发生,视具体情况而定)定义的主层(参见 A.3.2.). 下的主层策略)。
The population of patients targeted by the clinical question. This will be represented by the entire trial population, a subgroup defined by a particular characteristic measured at baseline, or a principal stratum defined by the occurrence (or non-occurrence, depending on context) of a specific intercurrent event (see Principal Stratum strategies under变量(或终点):为解决临床问题从每个患者获得的变量(或终点)。变量定义可能包括患者是否发生伴发事件(参见
A.3.2.).下的复合变量策略和在治策略)。
The其他伴发事件: variable (or endpoint) to be obtained for each patient that is required to address the clinical question. The specification of the variable might include whether the patient experiences an intercurrent event (see Composite Variable and While on Treatment strategies under A.3.2.). 在申办方与监管机构关于相关临床问题的交流中,治疗、人群和变量的精确说明有助于解决一些伴发事件。针对任何其他伴发事件的临床相关问题,通常采用疗法策略、假想策略或在治策略来反映。
Precise specifications of treatment, population and variable are likely to address many of the intercurrent events considered in sponsor and regulator discussions of the clinical question of interest. The clinical question of interest in respect of any other intercurrent events will usually be reflected using the strategies introduced as treatment policy, hypothetical or while on treatment. 群体层面汇总:最后,应规定变量的群体层面的汇总统计量,为不同治疗之间的比较提供基础。
Finally, a population-level summary for the variable should be specified, providing a basis for comparison between treatment conditions.
When defining a treatment effect of interest, it is important to ensure that the definition identifies an effect due to treatment and not due to potential confounders such as differences in duration of observation or patient characteristics.在定义治疗效应时,重要的是能够明确效应是由治疗引起的,而不是由潜在的混杂因素如观察期或患者特征的差异等引起的。
A.3.4. Considerations for Constructing an Estimand构建估计目标的考量
The clinical questions of interest and associated estimands should be specified at the initial stages of planning any clinical trial. Precise specification of objectives for most trials will need to reflect discontinuation of treatment and use of additional or alternative treatments. In some settings terminal events, such as death, should be addressed. Some trial objectives can only be described with reference to clinical events, for example the duration of response in subjects who achieve a response.临床问题及与之相关联的估计目标,应当在计划临床试验的初始阶段予以明确。大多数临床试验目的的精确说明,需要体现终止治疗、使用额外治疗或其他治疗的影响。在某些情况下,还应说明死亡一类的终末事件。有些试验目的只能参照临床事件来描述,例如获得应答的受试者的应答持续时间。
The construction of an estimand should consider what is of clinical relevance for the particular treatment in the particular therapeutic setting. Considerations include the disease under study, the clinical context (e.g. the availability of alternative treatments), the administration of treatment (e.g. one-off dosing, short-term treatment or chronic dosing) and the goal of treatment (e.g. prevention, disease modification, symptom control). Also important is whether an estimate of the treatment effect can be derived that is reliable for decision making. For example, a clinical question on the treatment effect on clinical outcome regardless of which other therapies are to be used before that outcome is experienced differs to a clinical question on the treatment effect had no additional medication been available. Depending on the setting, either might represent a clinical question of interest. However, in both cases, a clinical trial designed to estimate these treatment effects will often include the possibility to use additional medications if medically required. For the former question, values after the use of additional treatment will be relevant. For the latter question, values after the additional treatment are not directly relevant since the values also reflect the impact of that additional medication. It should be agreed that reliable estimation is possible before the choice of estimand is finalised. This includes, for the latter question, the methods to replace observations that are not to be used in the analysis. 构建一个估计目标,应该考虑在特定医疗环境下特定治疗的临床相关性。需考虑的因素包括:所研究的疾病、临床情况(例如可供选择的其他治疗)、治疗方式(例如一次性给药、短期治疗或长期给药)和治疗目的(例如预防、疾病改善、症状控制)。同样重要的是,能否估计出可靠的治疗效应供决策之用。例如,在临床结局发生前,无论是否使用其他治疗情况下的治疗效应和假设没有额外药物可用情况下的治疗效应,是不同的,它们可能都是值得关注的临床问题。但是,在这两种情况下,为估计这些治疗效应,相应的临床试验设计通常会考虑到在医学上需要使用额外药物的可能性。对于前一问题,使用额外治疗后的观测值是有意义的。对于后一问题,额外治疗后的观测值则无直接相关性,因为这些数值也反映了额外治疗的影响。在估计目标最终确定之前,应确认能够得到可靠的估计,包括后一个问题中,用什么方法替代未被分析使用的观测值。
When constructing the estimand it is necessary to have a clear understanding of the treatment to which the clinical question of interest pertains (see在构建估计目标时,有必要清楚地了解相关临床问题所涉及的治疗(见 A.3.3.).)。对治疗的明确说明可能已经反映了多个相关的伴发事件。具体而言,治疗可能已经反映了临床关注问题所涉及的以下变化:基础治疗、合并用药、使用额外或后线治疗、转组治疗和预处理方案。例如,可以将治疗指定为干预 ClearA加基础治疗B,并按需给药。这种情况下,无需将基础治疗B剂量的变化视为伴发事件。但是,需要将额外治疗视为伴发事件。如果治疗还涉及额外药物的使用,例如在使用疗法策略时,可将治疗定义为干预A加基础治疗B,按需给药,并按需使用额外药物。或者,如果治疗定义为干预 specifications for the treatments of interest might already reflect multiple relevant intercurrent events. Specifically, a treatment might already reflect the clinical question of interest in respect of changes in background treatment, concomitant medications, use of additional or later-line therapies, treatment-switching and conditioning regimens. For example, it is possible to specify treatment as intervention A added to background therapy B, dosed as required. In that case, changes to the dose of background therapy B would not need to be considered as an intercurrent event. However, the use of an additional therapy would need to be considered as an intercurrent event. If use of any additional medication is also reflected, using the treatment policy strategy for example, then treatment might be specified as intervention A added to background therapy B, dosed as required, and with additional medication, as required. Alternatively, if the treatment is specified as intervention A, then both changes in background therapy and use of additional therapy would be addressed as intercurrent events.A,那么基础治疗的变化和额外治疗的使用都将视为伴发事件。
Discussions should also consider whether specifications for the population and variable attributes should be used to reflect the clinical question of interest in respect of any intercurrent events. Strategies can then be considered for any other intercurrent events. Usually an iterative process will be necessary to reach an estimand that is of clinical relevance for decision making, and for which a reliable estimate can be made. Some estimands, in particular those for which the measurements taken are relevant to the clinical question, can often be robustly estimated making few assumptions. Other estimands may require methods of analysis with more specific assumptions that may be more difficult to justify and that may be more sensitive to plausible changes in those assumptions (see还应讨论是否通过明确人群和变量属性来说明相关临床问题的伴发事件。然后可以考虑有关任何其他伴发事件的策略。通常需要反复讨论来确定对于决策具有临床相关性,并且能得出可靠估计值的估计目标。一些估计目标,特别是那些观测值与临床问题相关的估计目标,通常可以通过很少的假设做出稳健的估计。而有些估计目标的分析方法可能需要更具体的假设,这些假设可能更难以论证,并且可能对假设的合理变化更敏感(见
A.5.1.). Where significant issues exist to develop an appropriate trial design or to derive an adequately reliable estimate for a particular estimand, an alternative estimand, trial design and method of analysis would need to be considered.)。对于某一特定的估计目标,如果在试验设计的合理性或者估计值的可靠性方面存在明显不足的话,就需要考虑另一种估计目标、试验设计和分析方法。
Avoiding or over-simplifying the process of discussing and constructing an estimand risks misalignment between trial objectives, trial design, data collection and method of analysis. Whilst an inability to derive a reliable estimate might preclude certain choices of strategy, it is important to proceed sequentially from the trial objective and an understanding of the clinical question of interest, and not for the choice of data collection and method of analysis to determine the estimand.省略或过度简化讨论和构建估计目标的过程,会产生导致试验目的、试验设计、数据收集和分析方法之间不一致的风险。当无法得出可靠的估计值时,可能会妨碍某些策略的选择,重要的是要从试验目的和对临床相关问题的理解出发,而不是为了选择数据收集和分析方法,来确定估计目标。
The experimental situation should also be considered. If the management of subjects (e.g. dose adjustment for intolerance, rescue treatment for inadequate response, burden of clinical trial assessments) under a clinical trial protocol is justified to be different to that which is anticipated in clinical practice, this might be reflected in the construction of the estimand.还应考虑试验现状。如果根据临床试验方案对受试者的管理(例如,因不耐受而进行剂量调整,因应答不足而做的补救治疗,临床试验评估所带来的负担)被证实与临床实践中所预期的不同,这可能要在估计目标的构建中有所体现。
Once constructed, the estimand should define a target of estimation clearly and unambiguously. Consider an intercurrent event of discontinuation of treatment; it is of utmost importance to distinguish between treatment effects of interest based on the principal stratum of patients who would be able to continue if administered the test treatment and the effect during continued treatment. Furthermore, neither of these should be taken to represent an effect if all patients can continue with treatment. 估计目标的构建,应该明确和清晰地定义估计的目标。以终止治疗伴发事件为例,最重要的是区分基于假设接受试验药物就能继续治疗的患者主层的治疗效应与实际持续治疗期间的效应。此外,如果所有患者都能继续治疗,则这两种情况都不能用来反映效应。
As stated above, when using the hypothetical strategy, some conditions are likely to be more acceptable for regulatory decision making than others. The hypothetical conditions described should therefore be justified for the quantification of an interpretable treatment effect that is relevant to inform the decisions to be taken by regulators, and use of the medicine in clinical practice. The question of what the values for the variable of interest would have been if rescue medication had not been available may be an important one. In contrast, the question of what the values for the variable of interest would have been under the hypothetical condition that subjects who discontinued treatment because of adverse drug reaction had in fact continued with treatment, might not be justifiable as being of clinical or regulatory interest. A clinical question of interest based on the effect if all subjects had been able to continue with treatment is not well-defined without a thorough discussion of the hypothetical conditions under which it is supposed that they would have continued. The inability to tolerate a treatment may constitute, in itself, evidence of an inability to achieve a favourable outcome. 如上所述,当使用假想策略时,某些情形更可能为监管决策所接受。因此,所描述的假想情形应该能合理地用来量化可解释的治疗效应,为监管机构做决策和临床实践中药物的使用提供相关信息。假如未获得补救药物,变量值会是多少也许是一个重要问题。相反,如果假想情形中因药物不良反应而终止治疗的受试者实际上继续接受治疗,那么相关变量值在这一假想情形下会是多少这个问题可能不具有临床或监管意义。假如所有受试者都能够继续接受治疗,但没有对他们会继续这一假想情形进行充分讨论的话,则基于该效应的临床问题的定义是不充分的。药物不耐受本身可能就构成了无法达到有利结局的证据。
Characterising beneficial effects using estimands based on the treatment policy strategy might also be more generally acceptable to support regulatory decision making, specifically in settings where estimands based on alternative strategies might be considered of greater clinical interest, but main and sensitivity estimators cannot be identified that are agreed to support a reliable estimate or robust inference. An estimand based on the treatment policy strategy might offer the possibility to obtain a reliable estimate of a treatment effect that is still relevant. In this situation, it is recommended to also include those estimands that are considered to be of greater clinical relevance and to present the resulting estimates along with a discussion of the limitations, in terms of trial design or statistical analysis, for that specific approach. When constructing estimands based on the treatment policy strategy, inference can be complemented by defining an additional estimand and analysis pertaining to each intercurrent event for which the strategy is used; for example, contrasting both the treatment effect on a symptom score and the proportion of subjects using additional medication under each treatment. Similarly, an estimand using a while on treatment strategy should usually be accompanied by the additional information on the time to intercurrent event distributions, and an estimand based on a principal stratum would usefully be accompanied by information on the proportion of patients in that stratum, if available. 使用基于疗法策略的估计目标来描述获益效应以支持监管决策,可能更被普遍接受,特别是在某些情况下,尽管基于其他策略的估计目标可能被认为更具临床意义,但是无法找到其公认的能支持可靠估计值或稳健推断的主估计方法和敏感性估计方法。基于疗法策略的估计目标仍然可能得到具有临床相关性的可靠估计值。在这种情况下,建议还包括那些被认为具有更大临床相关性的估计目标,并给出所得到的估计值,以及关于该特定方法在试验设计或统计分析方面的局限性的讨论。在构建基于疗法策略的估计目标时,可以对使用该策略的每个伴发事件定义额外的估计目标和分析来补充推断;例如,对比各治疗组中治疗对症状评分的治疗效应和使用额外药物的受试者比例。类似的,使用在治策略的估计目标通常应该有伴发事件发生时间分布的附加信息,基于主层策略的估计目标通常有关于该主层中患者比例的信息(如果有)。
The基于非劣效性或等效性目的构建估计目标以支持监管决策的考虑,可能与基于优效性目的的估计目标不同。正如在ICH considerations informing the construction of estimand to support regulatory decision making based on a non-inferiority or equivalence objective may differ to those for the choice of estimand for a superiority objective. As explained in ICH E9, the problem facing the regulator in their decision making is different when based on non-inferiority or equivalence studies compared to superiority studies. In SectionE9中所解释的,非劣效性或等效性研究与优效性研究相比,监管机构在决策中面临的问题是不同的。在第 3.3.2. it is stated that such trials are not conservative in nature and the importance of minimising the number of protocol violations and deviations, non-adherence and study withdrawals is indicated. In Section 5.节中指出,这类试验本质上不保守,重要的是尽量减少方案违背和偏离、不依从和退出研究的数量。在第5.2.1. it is described that the result of the Full Analysis Set (FAS) is generally not conservative and that its role in such trials should be considered very seriously. Estimands that are constructed with one or more intercurrent events accounted for using the treatment policy strategy present similar issues for non-inferiority and equivalence trials as those related to analysis of the FAS under the ITT principle. Responses in both treatment groups can appear more similar following discontinuation of randomised treatment or use of another medication for reasons that are unrelated to the similarity of the initially randomised treatments. Estimands could be constructed to directly address those intercurrent events which can lead to the attenuation of differences between treatment arms (e.g. discontinuations from treatment and use of additional medications). When selecting strategies, it might be important to distinguish between trials designed to detect whether differences exist between treatments containing the same or similar active substance (e.g. comparison of a biosimilar to a reference treatment) and trials where a non-inferiority or equivalence hypothesis is used in order to establish and quantify evidence of efficacy. An estimand can be constructed to target a treatment effect that prioritises sensitivity to detect differences between treatments, if appropriate for regulatory decision making.节中指出,全分析集(FAS)的结果一般不是保守的,它在这类试验中的作用应该被认真考虑。使用疗法策略来说明由一个或多个伴发事件构建的估计目标,对于非劣效性和等效性试验而言,存在的问题与ITT原则下使用FAS分析存在的相关问题类似。终止随机分配的治疗或因各种原因使用另一种药物,两个治疗组的应答情况可以表现得更相近,而这与最初的随机分配的治疗组间的相似度无关。可以构建估计目标来直接说明那些可能导致治疗组之间差异被弱化的伴发事件(例如终止治疗和使用额外药物)。在选择策略时,区分用于检测出含有相同或相似药物活性成分的治疗之间是否有差异的试验(例如,将生物类似物与参比治疗进行比较)与采用非劣效性或等效性假设来建立和量化有效性证据的试验是很重要的。为适用于监管决策,可以构建一个针对治疗效应的估计目标,优先考虑能更灵敏地检测出治疗间效应的差异。
A.4. IMPACT ON TRIAL DESIGN AND CONDUCT对试验设计和实施的影响
The design of a trial needs to be aligned to the estimands that reflect the trial objectives. A trial design that is suitable for one estimand might not be suitable for other estimands of potential importance. Clear definitions for the estimands on which quantification of treatments effects will be based should inform the choices that are made in relation to trial design. This includes determining the inclusion and exclusion criteria that identify the target population, the treatments, including the medications that are allowed and those that are prohibited in the protocol, and other aspects of patient management and data collection. If interest lies, for example, in understanding the treatment effect regardless of whether a particular intercurrent event occurs, a trial in which the variable is collected for all subjects is appropriate. Alternatively, if the estimands that are required to support regulatory decision making do not require the collection of the variable after an intercurrent event, then the benefits of collecting such data for other estimands should be weighed against any complications and potential drawbacks of the collection. 试验设计需要与反映试验目的的估计目标相一致。一种适用于某个估计目标的试验设计,不一定适用于其他具有潜在重要性的估计目标。治疗效应的量化依赖于估计目标,而估计目标的明确定义应当为试验设计的选择提供相关信息。这包括定义目标人群的入选和排除标准,治疗(包括方案中允许的药物和禁用药物),以及患者管理和数据收集等方面。例如,如果关注的是不管是否发生特定伴发事件的治疗效应,则该试验应收集所有受试者的变量。或者,如果用于支持监管决策而制定的估计目标不需要收集伴发事件后的变量值,则应权衡为其他估计目标收集此类数据的益处与数据收集的复杂性和潜在缺陷。
Efforts should be made to collect all data that are relevant to support estimation, including data that inform the characterisation, occurrence and timing of intercurrent events. Data cannot always be collected. Certainly, subjects cannot be retained in a trial against their will, and in some trials missing data for some subjects is inevitable by design, such as administrative censoring in trials with survival outcomes. On the contrary, the occurrence of intercurrent events such as discontinuation of treatment, treatment switching, or use of additional medication, does not imply that the variable cannot be measured thereafter, though the measures may not be relevant. For terminal events such as death, the variable cannot be measured after the intercurrent event, but neither should these data generally be regarded as missing.应尽可能收集所有与估计相关的数据,包括伴发事件的特征、发生情况和时间。然而数据不是总能被收集到,不能违背受试者意愿将其留在试验中,而且在某些试验中,受试者数据的缺失按照设计是不可避免的,如生存结局试验中的管理性删失。相反,如发生终止治疗、转组治疗或使用额外药物等伴发事件,尽管这些测量结果可能并不相关,并不意味着在事件之后无法测量变量。对于死亡等终末事件,变量不能在伴发事件后进行观测,但这些数据通常都不应被视为缺失。
Not collecting any data needed to assess an estimand results in a missing data problem for subsequent statistical inference. The validity of statistical analyses may rest upon untestable assumptions and, depending on the proportion of missing data, this may undermine the robustness of the results (see不收集评估估计目标所需的数据,将导致后续统计推断中的缺失数据问题。统计分析的合理性可能取决于不可验证的假设和缺失数据的比例,这些可能会削弱结果的稳健性(见 A.5.). A prospective plan to collect informative reasons for why data intended for collection are missing may help to distinguish the occurrence of intercurrent events from missing data. This in turn may improve the analysis and may also lead to a more appropriate choice of sensitivity analysis. For example, “loss to follow-up” may more accurately be recorded as “treatment discontinuation due to lack of efficacy”. Where that has been defined as an intercurrent event, this can be reflected through the strategy chosen to account for that intercurrent event and not as a missing data problem. To reduce missing data, measures can be implemented to retain subjects in the trial. However, measures to reduce or avoid intercurrent events that would normally occur in clinical practice risk reducing the external validity of the trial. For example, selection of the trial population or use of titration schemes or concomitant medications to mitigate the impact of toxicity might not be suitable if those same measures would not be implemented in clinical practice. )。制定一个前瞻性计划收集详细的数据缺失的原因,将有助于区分伴发事件的发生与缺失数据。这样可改进分析,并可使敏感性分析的选择更合理。例如,将“失访”记录为“因缺乏疗效而终止治疗”可能更准确。在将其定义为伴发事件的情况下,可以选择相应策略,而不是将其作为缺失数据问题来处理。为减少缺失数据,可采取措施将受试者保留在试验中。然而,减少或避免临床实践中通常会发生的伴发事件的措施存在降低试验外部有效性的风险。例如,如果在临床实践中不会通过选择试验人群,或使用滴定方案,或使用合并用药来减轻毒性的影响,那么在试验中采取这些措施也就不合适了。
Randomisation and blinding remain cornerstones of controlled clinical trials. Design techniques for avoiding bias are addressed in Section随机化和盲法仍然是对照临床试验的基石。避免偏倚的设计方法请见第 2.3. Certain estimands may necessitate, or may benefit from, use of trial designs such as run-in or enrichment designs, randomised withdrawal designs, or titration designs. It might be of interest to identify the principal stratum of subjects who can tolerate a treatment using a run-in period, in advance of randomising those subjects between test treatment and control. Dialogue between regulator and sponsor would need to consider whether the proposed run-in period is appropriate to identify the target population, and whether the choices made for the subsequent trial design (e.g. washout period, randomisation) supports the estimation of the target treatment effect and associated inference. These considerations might limit the use of these trial designs, and use of that particular strategy.节。某些估计目标可能需要或受益于试验设计的运用,如导入期或富集设计、随机撤药设计或滴定设计。在对受试者进行随机化分组之前,利用导入期识别对药物耐受的受试者主层,这个办法是可取的。监管机构和申办方之间的交流需要考虑拟定的导入期是否适合于确定目标人群,以及后续试验设计(例如,洗脱期、随机化)的选择是否支持目标治疗效应的估计和相关推断。这些考虑可能会限制这些试验设计的使用,以及特定策略的使用。
A precise description of the treatment effects of interest should inform sample size calculations. Particular care should be taken when making reference to historical studies that might, implicitly or explicitly, have reported estimated treatment effects or variability based on a different estimand. Where all subjects contribute information to the analysis, and where the impact of the strategy to reflect intercurrent events is included in the effect size that is targeted and the expected variance, it is not usually necessary to additionally inflate the calculated sample size by the expected proportion of subject withdrawals from the trial.对治疗效应的精确描述应当为样本量计算提供信息。在参照历史研究时应特别谨慎,这些历史研究可能隐含或明确地报告了基于不同估计目标的治疗效应或变异的估计值。当所有受试者均能为分析提供信息,且在目标效应量和预期方差中已考虑了相应的策略来反映伴发事件的影响时,则在计算样本量时通常不需要按预期退出试验的受试者比例额外增加样本量。
Section第 7.2. addresses issues related to summarising data across clinical trials. The need to have consistent definitions for the variables of interest is highlighted and this can be extended to the construction of estimands. Hence, in situations when synthesising evidence from across a clinical trial programme is envisaged at the planning stage, a suitable estimand should be constructed, included in the trial protocols, and reflected in the choices made for the design of the contributing trials. Similar considerations apply to the design of a meta-analysis, using estimated effect sizes from completed trials to determine non-inferiority margins, or the use of
external control groups for the interpretation of single-arm trials. A naïve comparison between data sources, or integration of data from multiple trials without consideration and specification of the estimand that is addressed in each data presentation or statistical analysis, could be misleading. 节说明了关于跨临床试验数据汇总的问题。强调对所关注的变量应有一致的定义,并且这可延伸用于估计目标的构建。因此,为了从多个临床试验获得综合性证据,在计划阶段应构建一个合适的估计目标,使其包含在项目里的所有试验方案中,并反映在相关试验的设计选择中。类似的考虑适用于meta分析的设计,使用已完成的试验中估计的效应量来确定非劣效界值,或者使用外部对照组来诠释单臂试验。在没有考虑和说明每个数据呈现或统计分析所对应的估计目标的情况下,不同来源数据之间的简单比较,或者来自多个试验的数据整合可能会产生误导。
More generally, a trial is likely to have multiple objectives translated into multiple estimands, each associated with statistical testing and estimation. The multiplicity issues arising should be addressed.总的来说,一个试验有可能将多个目的转化为多个估计目标,每个估计目标都与统计检验和估计相关联。此时,应当考虑其中的多重性问题。
A.5. IMPACT ON TRIAL ANALYSIS对试验分析的影响
A.5.1. Main Estimation主要估计
An estimand for the effect of treatment relative to a control will be estimated by comparing the outcomes in a group of subjects on the treatment to those in a similar group of subjects on the control. For a given estimand, an aligned method of analysis, or estimator, should be implemented that is able to provide an estimate on which reliable interpretation can be based. The method of analysis will also support calculation of confidence intervals and tests for statistical significance. An important consideration for whether an interpretable estimate will be available is the extent of assumptions that need to be made in the analysis. Key assumptions should be stated explicitly together with the estimand and accompanying main and sensitivity estimators. Assumptions should be justifiable and implausible assumptions should be avoided. The robustness of the results to potential departures from the underlying assumptions should be assessed through an estimand-aligned sensitivity analysis (see对于治疗组相较于对照组的治疗效应,估计目标通过比较具有相似受试者的试验组和对照组的结果来进行估计。对于给定的估计目标,应采用与其相一致的分析方法(或估计方法),使所得估计值可以支持对结果的可靠解读。该分析方法还应能计算置信区间并进行统计学显著性的检验。可解释的估计值是否存在,一个重要的考虑因素是分析中需要作出的假设的程度。关键假设应与估计目标及其主要估计方法和敏感性估计方法一起明确说明。假设应是合理的,要避免不恰当的假设。对于潜在的偏离假设的情形,结果的稳健性应通过与估计目标相对应的敏感性分析进行评估(见 A.5.2.). Estimation that relies on many or strong assumptions requires more extensive sensitivity analysis. Where the impact of deviations from assumptions cannot be comprehensively investigated through sensitivity analysis, that particular combination of estimand and method of analysis might not be acceptable for decision making.)。如果一个估计依赖于多种假设或强假设,则需要更广泛的敏感性分析。如果偏离假设所造成的影响不能通过敏感性分析进行全面研究,那么这种特定的估计目标和分析方法的组合可能无法用于决策。
All methods of analysis rely on assumptions, and different methods may rely on different assumptions even when aligned to the same estimand. Nevertheless, some kinds of assumption are inherent in all methods of analysis aligned to estimands that use each of the different strategies outlined; for example, the methodology for predicting the outcomes that would have been observed in the hypothetical scenario, or for identifying a suitable target population in a principal stratum strategy. Some examples are given below related to the different strategies used to reflect the occurrence of intercurrent events. The issues highlighted will be key components of discussion between sponsor and regulator in advance of an estimand, main analysis and sensitivity analysis being agreed.所有的分析方法都依赖于假设,而且即使对应于相同的估计目标,不同的分析方法也可能依赖于不同的假设。尽管如此,对于使用每种不同策略的估计目标,相对应的所有方法中都存在一些固有的假设。例如,预测在假想情形下本可观察到的结局的方法,或者在主层策略下确定合适目标人群的方法。下面列举了一些关于针对不同伴发事件采用不同策略的例子。在申办方和监管机构就估计目标、主要分析和敏感性分析达成一致之前,这里所强调的问题将是双方讨论的关键。
Analysis aligned with a treatment policy strategy to address a given intercurrent event may entail stronger or weaker assumptions depending on the design and conduct of the trial. When most subjects are followed-up even after the respective intercurrent event (e.g. discontinuation of treatment), the remaining problem of missing data may be relatively minor. In contrast, when observation is terminated after an intercurrent event, which is obviously undesirable in respect of this strategy, the assumption that (unobserved) outcomes for discontinuing subjects are similar to the (observed) outcomes for those who remain on treatment will often be implausible. An alternative approach to handle the missing data would need to be justified and sensitivity analysis will be expected. 与针对某一伴发事件的疗法策略相对应的分析,根据试验的设计和实施,可能需要或强或弱的假设。如果在相应的伴发事件(如治疗终止)之后仍对大多数受试者进行随访,那么缺失数据问题可能相对较小。相反,如果伴发事件后停止观察(对于该策略显然是不提倡的),那么假设终止治疗受试者的(未观察到的)结局与继续治疗的受试者的(观察到的)结局相似通常是不合理的。此时在处理缺失数据时需要对其他方法进行论证,并进行敏感性分析。
Analysis aligned to a hypothetical strategy involves outcomes different from those actually observed; for example, outcomes if rescue medication had not been given when in fact it was. Observations before the rescue medication and observations on subjects who did not require rescue medication may be informative, but only under strong assumptions. 与假想策略相对应的分析方法所涉及的结局与实际观测的结局不同;例如,尽管实际上给予了补救药物,但需要估计未给予补救药物时的结局。在给予补救药物之前的观测值与不需要补救药物的受试者的观测值可能提供有效信息,但需要更强的假设。
A composite variable strategy can avoid statistical assumptions about data after an intercurrent event by considering occurrence of the intercurrent event as a component of the outcome. The potential concern relates less to assumptions for estimation, and more to the interpretation of the estimated treatment effect. For the estimand to be interpretable, if scores are assigned for failure because the intercurrent event occurs, these should meaningfully reflect the lack of benefit to the patient (e.g. death may be reflected differently than discontinuation of treatment due to adverse event). 复合变量策略可以通过将伴发事件的发生视为结局的组成部分来避免对伴发事件后的数据做出统计学假设。这种情况下,潜在的担忧往往不在于估计中相应的假设,而在于对治疗效应估计结果的解释。为使估计目标得以合理解释,如果将伴发事件的发生认定为失败,并给出评分,这个评分应该能够有效地反映出患者获益的缺乏程度(例如,对死亡与因不良事件导致处理终止的反映也许不同)。
Estimands constructed based on a while on treatment strategy can be estimated provided outcomes are collected up to the time of the intercurrent event. Again, the crucial assumptions concern interpretation. Take discontinuation of treatment by way of example. Outcomes while on treatment may be improved but the treatment may also shorten, or lengthen, the treatment period by provoking, or delaying, discontinuations, and both these effects should be considered in interpretation and assessment of clinical benefit.如果伴发事件发生之前的结局已被收集,那么依据在治策略所构建的估计目标就可以被估计。同样的,此处的关键假设将影响到结果的解释。以终止治疗为例,治疗过程中结局可能会改善,但同时治疗也可能因为引发、延迟、终止治疗等原因,使得治疗期缩短或延长。此类影响应在解释和评估临床获益时予以考虑。
Analysis aligned to a principal stratum strategy usually requires strong assumptions. For example, some principal stratification methods infer this from baseline characteristics of the subjects, but the correctness of this inference may be difficult to assess. This difficulty cannot be avoided by simplified methods, however. For example, simply comparing subjects who do not have an intercurrent event on the test treatment to those who do not have an event on control, assuming intercurrent events are unrelated to treatment, is very difficult to justify. 与主层策略相对应的分析通常需要较强假设。例如,一些主层方法是基于受试者的基线特征而推断出来的,但这种推断的正确性可能难以评估。而且,这种困难不能通过简化的方法来避免。例如,假设伴发事件与处理无关,并简单地比较试验组和对照组中未出现伴发事件的受试者,这是非常难以论证的。
Even after defining estimands that address intercurrent events in an appropriate manner and making efforts to collect the data required for estimation (see即使以恰当的方式定义了解决伴发事件的估计目标,并努力收集估计所需的数据(见 A.4.), some data may still be missing, including e.g. administrative censoring in trials with survival outcomes. Failure to collect relevant data should not be confused with the choice not to collect, or to collect and not to use, data made irrelevant by an intercurrent event. For example, data that were intended to be collected after discontinuation of trial medication to inform an estimand based on the treatment policy strategy are missing if uncollected; however, the same data points might be irrelevant for another strategy, and thus, for the purpose of that second estimand, are not missing if uncollected. Where those efforts to collect data are not successful it becomes necessary to make assumptions to handle the missing data in the statistical analysis. Handling of missing data should be based on clinically plausible assumptions and, where possible, guided by the strategies employed in the description of the estimand. The approach taken may be based on observed covariates and post-baseline data from individual subjects and from other similar subjects. Criteria to identify similar subjects might include whether or not the intercurrent event has occurred. For example, for subjects who discontinue treatment without further data being collected, a model may use data from other subjects who discontinued treatment but for whom data collection has continued.),一些数据仍然可能缺失。例如,生存结局试验中的管理性删失。未能收集到相关数据不应与选择不收集或选择收集但不使用(因伴发事件变得无关的数据)相混淆。例如,对于基于疗法策略的估计目标,在终止试验药物后的数据仍应被收集,如果未收集,则视为数据缺失;然而,对另一种策略而言,相同的数据点可能不相关,因此,对于相应的估计目标,此类未收集数据则不会被视为缺失。如果数据收集不完整,则有必要在统计分析中对缺失数据的处理做出一些假设。缺失数据的处理应基于临床上的合理假设,并在可能的情况下以估计目标描述中采用的策略为指导。采取的方法可能基于个体受试者和与其相似受试者所观测到的协变量和基线后数据。识别相似受试者的标准可能包括是否发生伴发事件。例如,对于终止治疗但未收集更多数据的受试者,可使用终止治疗但继续收集数据的其他受试者的数据来建模。
A.5.2. Sensitivity Analysis 敏感性分析
A.5.2.1. Role of Sensitivity Analysis 敏感性分析的作用
Inferences based on a particular estimand should be robust to limitations in the data and deviations from the assumptions used in the statistical model for the main estimator. This robustness is evaluated through a sensitivity analysis. Sensitivity analysis should be planned for the main estimators of all estimands that will be important for regulatory decision making and labelling in the product information. This can be a topic for discussion and agreement between sponsor and regulator. 基于特定估计目标的统计推断,应该对数据的局限以及主估计方法统计模型中假设的偏离具有稳健性。这种稳健性应通过敏感性分析来评价。对于所有用于监管决策和说明书制定的估计目标的主估计方法,都应有相应的敏感性分析计划。此问题需要在申办方和监管机构之间讨论并达成一致。
The statistical assumptions that underpin the main estimator should be documented. One or more analyses, focused on the same estimand, should then be pre-specified to investigate these assumptions with the objective of verifying whether or not the estimate derived from the main estimator is robust to departures from its assumptions. This might be characterised as the extent of departures from assumptions that change the interpretation of the results in terms of their statistical or clinical significance (e.g. tipping point analysis).支持主估计方法的统计假设应明确记录。对于同一估计目标,应该预先规定一项或多项分析来评估这些假设,目的是验证根据主估计方法得出的估计值是否对假设偏离具有稳健性。其衡量标准可以是对假设不同程度的偏离是否会改变结果的统计学或临床意义(如临界点分析)。
Distinct from sensitivity analysis, where investigations are conducted with the intent of exploring robustness of departures from assumptions, other analyses that are conducted in order to more fully investigate and understand the trial data can be termed “supplementary analysis” (see Glossary; 敏感性分析旨在探索偏离假设时分析结果的稳健性,与此不同的,为了更全面地研究和理解试验数据而进行的其他分析可称为“补充分析”(见词汇表,A.5.3.). Where the primary estimand(s) of interest is agreed between sponsor and regulator, the main estimator is pre-specified unambiguously, and the sensitivity analysis verifies that the estimate derived is reliable for interpretation, supplementary analyses should generally be given lower priority in assessment.)。如果申办方和监管机构就所关注的主要估计目标达成一致,并预先明确规定了主估计方法,且敏感性分析也验证了估计值的结果解释是可靠的,则补充分析在结果评估中通常不被优先考量。
A.5.2.2. Choice of Sensitivity Analysis敏感性分析的选择
When planning and conducting a sensitivity analysis, altering multiple aspects of the main analysis simultaneously can make it challenging to identify which assumptions, if any, are responsible for any potential differences seen. It is therefore desirable to adopt a structured approach, specifying the changes in assumptions that underlie the alternative analyses, rather than simply comparing the results of different analyses based on different sets of assumptions. The need for analyses varying multiple assumptions simultaneously should then be considered on a case by case basis. A distinction between testable and untestable assumptions may be useful when assessing the interpretation and relevance of different analyses.当计划和实施敏感性分析时,同时改变主要分析的多个方面可能难以确定由哪些假设导致了目前所观测到的潜在差异。因此,通常采用结构化的方法,指定不同分析背后的假设的变化,而不是简单地基于一组不同的假设比较不同分析的结果。应根据具体情况考虑是否需要同时改变多个假设的分析。在评估不同分析的解释和相关性时,区分可验证的和不可验证的假设可能是有帮助的。
The need for sensitivity analysis in respect of missing data is established and retains its
importance in this framework. Missing data should be defined and considered in respect of a
particular estimand (see在本文所设立框架中,进一步明确了对缺失数据进行敏感性分析的必要性和重要性。缺失数据应依据特定估计目标进行定义和考虑(见 A.4.). The distinction between data that are missing in respect of a
specific estimand and data that are not directly relevant to a specific estimand gives rise to
separate sets of assumptions to be examined in sensitivity analysis.)。对应于特定估计目标的缺失数据,以及与特定估计目标不直接相关的数据,两者之间存在区别,由此在分析中产生了不同类别的假设,需要通过敏感性分析来检查。
A.5.3. Supplementary Analysis补充分析
Interpretation of trial results should focus on the main estimator for each agreed estimand providing that the corresponding estimate is verified to be robust through the sensitivity analysis. Supplementary analyses for an estimand can be conducted in addition to the main and sensitivity analysis to provide additional insights into the understanding of the treatment effect. They generally play a lesser role for interpretation of trial results. The need for, and utility of, supplementary analyses should be considered for each trial. 试验结果的解释应侧重于对应每个估计目标的主估计方法,并通过敏感性分析验证相应估计值的稳健性。除了主要分析和敏感性分析之外,还可以对估计目标进行补充分析,以提供对治疗效应更全面的了解。补充分析在解释试验结果方面的作用通常较小。每项试验均需考虑补充分析的必要性和作用。
Section第 5.2.3. indicates that it is usually appropriate to plan for analyses based on both the FAS and the Per Protocol Set (PPS) so that differences between them can be the subject of explicit discussion and interpretation. Consistent results from analyses based on the FAS and the PPS is indicated as increasing confidence in the trial results. It is also described in Section 5.节指出,同时基于全分析集(FAS)和符合方案集(PPS)的分析计划通常是适当的,从而它们之间的差异会成为讨论和结果解读的关键。如果基于FAS分析和PPS分析的结果一致,则可增强试验结果的可信度。第5.2.2. that results based on a PPS might be subject to severe bias. In respect of the framework presented in this addendum, it may not be possible to construct a relevant estimand to which analysis of the PPS is aligned. As noted above, analysis of the PPS does not achieve the goal of estimating the effect in any principal stratum, for example, in those subjects able to tolerate and continue to take the test treatment, because it may not compare similar subjects on different treatments.节还指出,基于PPS的结果可能会产生严重偏倚。就本增补中提出的框架而言,可能无法构建与PPS分析相对应的估计目标。如上所述,PPS分析不能实现在任何主层(例如,在能够耐受并继续接受试验药物的受试者)中估计效应的目的,因为PPS所比较的受试者在不同治疗组之间可能不具有可比性。
Protocol violations and deviations might exclude subjects from the PPS, for example by having a visit outside a time window, without an intercurrent event necessarily having occurred. Likewise, subjects could experience an intercurrent event, such as death, without having deviated from the protocol. Notwithstanding the differences between violations and deviations from the protocol and intercurrent events, events likely to affect the interpretation or existence of measurements are considered in the description of the estimand. Estimands might be
constructed, with aligned method of analysis, that better address the objective usually associated with the analysis of the PPS. If so, analysis of the PPS might not add additional insights.即使没有发生伴发事件,方案违背和偏离(例如,在时间窗外进行访视)也可能会使受试者从PPS中被排除。同样,受试者可能发生伴发事件(例如死亡)但却没有偏离方案。尽管违背和偏离方案与伴发事件之间存在差异,在估计目标的描述中仍应考虑可能影响观测结果解释或存在的事件。通过构建估计目标和相应的分析方法,可能更好地反映与PPS分析相关的目标。此时,PPS分析也许不能提供额外的信息。
A.6. DOCUMENTING ESTIMANDS AND SENSITIVITY ANALYSIS估计目标和敏感性分析的记录
A trial protocol should define and specify explicitly a primary estimand that corresponds to the primary trial objective. The protocol and the analysis plan should pre-specify the main estimator that is aligned with the primary estimand and leads to the primary analysis, together with a suitable sensitivity analysis to explore the robustness under deviations from its assumptions. Estimands for secondary trial objectives (e.g. related to secondary variables) that are likely to support regulatory decisions should also be defined and specified explicitly, each with a corresponding main estimator and a suitable sensitivity analysis. Additional exploratory trial objectives may be considered for exploratory purposes, leading to additional estimands.试验方案应当定义并明确说明与主要试验目的相对应的主要估计目标。方案和分析计划中应预先规定与主要估计目标一致并对应主要分析的主估计方法,以及当假设偏离时用来探索结果稳健性的合适的敏感性分析。对于可能支持监管决策的次要试验目的(例如,与次要变量相关),相应的估计目标也应当明确定义和说明,并且每个估计目标都有相应的主估计方法和合适的敏感性分析。还可以考虑属于探索性质的额外的探索性试验目的,此时也会产生额外的估计目标。
The choice of the primary estimand will usually be the main determinant for aspects of trial design, conduct and analysis. Following usual practices, these aspects should be well documented in the trial protocol. If secondary estimands are of key interest, these considerations may be extended to support these as needed and should be documented as well. Beyond these aspects, the conventional considerations for trial design, conduct and analysis remain the same.主要估计目标的选择通常是试验设计、实施和分析的主要决定因素。按照常规,这些信息应在试验方案中详细记录。如果次要估计目标同样需要重点关注,这些考虑可扩展到相应的估计目标,并同样在方案中记录。除此之外,对于试验设计、实施和分析的常规考虑仍然保持不变。
While it is to the benefit of the sponsor to have clarity on what is being estimated, it is not a regulatory requirement to document an estimand for each exploratory objective. 尽管明确阐明估计的内容对申办方有益,但监管部门并不要求对每一个探索性目的的估计目标都进行记录。
Results from the main, sensitivity and supplementary analyses should be reported systematically in the clinical trial report, specifying whether each analysis was pre-specified, introduced while the trial was still blinded, or performed post hoc. Summaries of the number and timings of each intercurrent event in each treatment group should be reported.在临床试验报告中应系统报告主要分析、敏感性分析和补充分析的结果,同时详细说明每项分析是否为预先规定的、在试验仍处于盲态时引入进行的,还是事后进行的。应汇总报告各处理组中各类伴发事件的数量和出现时间。
Changes to the estimand during the trial can be problematic and can reduce the credibility of the trial. Addressing intercurrent events that were not foreseen at the design stage, and are identified during the conduct of the trial, should discuss not only the choices made for the analysis, but the effect on the estimand, i.e. on the description of the treatment effect that is being estimated, and the interpretation of the trial results. A change to the estimand should usually be reflected through amendment to the protocol.试验期间改变估计目标可能是有问题的,这样做会降低试验的可信度。对于在设计阶段未预见但在试验实施过程中发现的伴发事件,不仅要讨论分析方法的选择,还要讨论它们对估计目标的影响,即对所估计的治疗效应描述的影响,和对试验结果解释的影响。估计目标的改变通常应通过修订试验方案来体现。
